|
This is the order for the question and the answer for it. The periodic table is arranged according to the periodic law. The elements in the modern table are arranged in order. He returned the electra negativity which is what decreases? Which is your lead's decrease? Here the question is asked, we write the order. Similar to atomic radius, the ionic radius of an element increases in size down a group as. Mendeleev's table did not arrange the elements according to recurring trends in their properties. When we go through the group of redwoods, we know they looked at the increase in local negative vibes. You're actually electra negative when you're along the period of electro negativity. 9) Give the name the element found at each of the. Which is equal to the electoral negativity of K. 6) Arrange the following elements in order of decreasing atomic size. R B's electoral negativity was mentioned. Electronegativity is the tendency of atoms to attract electrons. Which is 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 88 You're the value of R B, so the increasing order is equal to okay. The electronegativity values increase from left to right and bottom to top in the periodic table excluding the Noble gases. Electron affinity can be thought of as how much an element wants electrons. He looked at the order of the negative things. So you are prepared? Ready Look Trow was looking at something negative. These are electro negativities of the listed elements. Based on the values above, we can clearly arrange them. Once we are aware of the value of electro negativities for the element, we can organize them according to their values. The electoral order is written in the increasing order of the compound. To arrange them according to the order of electronegativity, we can employ two strategies. Press esc, or click the close the button to close this dialog box.Greetings students. Rank these elements according to the radii of their atoms, from smallest to. Search (Combination + S): Shortcut for search page. + H :Cl hydrogen chloride s&0 If the electronegativity difference. Site Map (Combination + M): Shortcut for site map (footer agency) section of the page. Group of answer choices Ionization energy Electronegativity Atomic radius None. Main Content (Combination + R): Shortcut for viewing the content section of the current page.įAQ (Combination + Q): Shortcut for FAQ page.Ĭontact (Combination + C): Shortcut for contact page or form inquiries.įeedback (Combination + K): Shortcut for feedback page. Solution for Arrange the following ions in order of increasing ionic radius: calcium ion, potassium ion. Home Page (Combination + H): Accessibility key for redirecting to homepage. Shortcut Keys Combination Activation Combination keys used for each browser.Ĭhrome for Linux press (Alt+Shift+shortcut_key)Ĭhrome for Windows press (Alt+shortcut_key)įor Firefox press (Alt+Shift+shortcut_key)įor Internet Explorer press (Alt+Shift+shortcut_key) then press (enter)Īccessibility Statement (Combination + 0): Statement page that will show the available accessibility keys. A guide to understanding and implementing Web Content Accessibility Guidelines 2.0 is available at: Compliance to these criteria is measured in three levels: A, AA, or AAA.  
There are testable success criteria for each guideline. Complete the Lewis dot (electrondot) structure for the given neutral atoms. Which of these elements has the highest first ionization energy chlorine. Largest - smallest radius Li Be B C N O F Ne. 
Arrange the elements according to atomic radius. Question: Arrange these elements according to electronegativity. 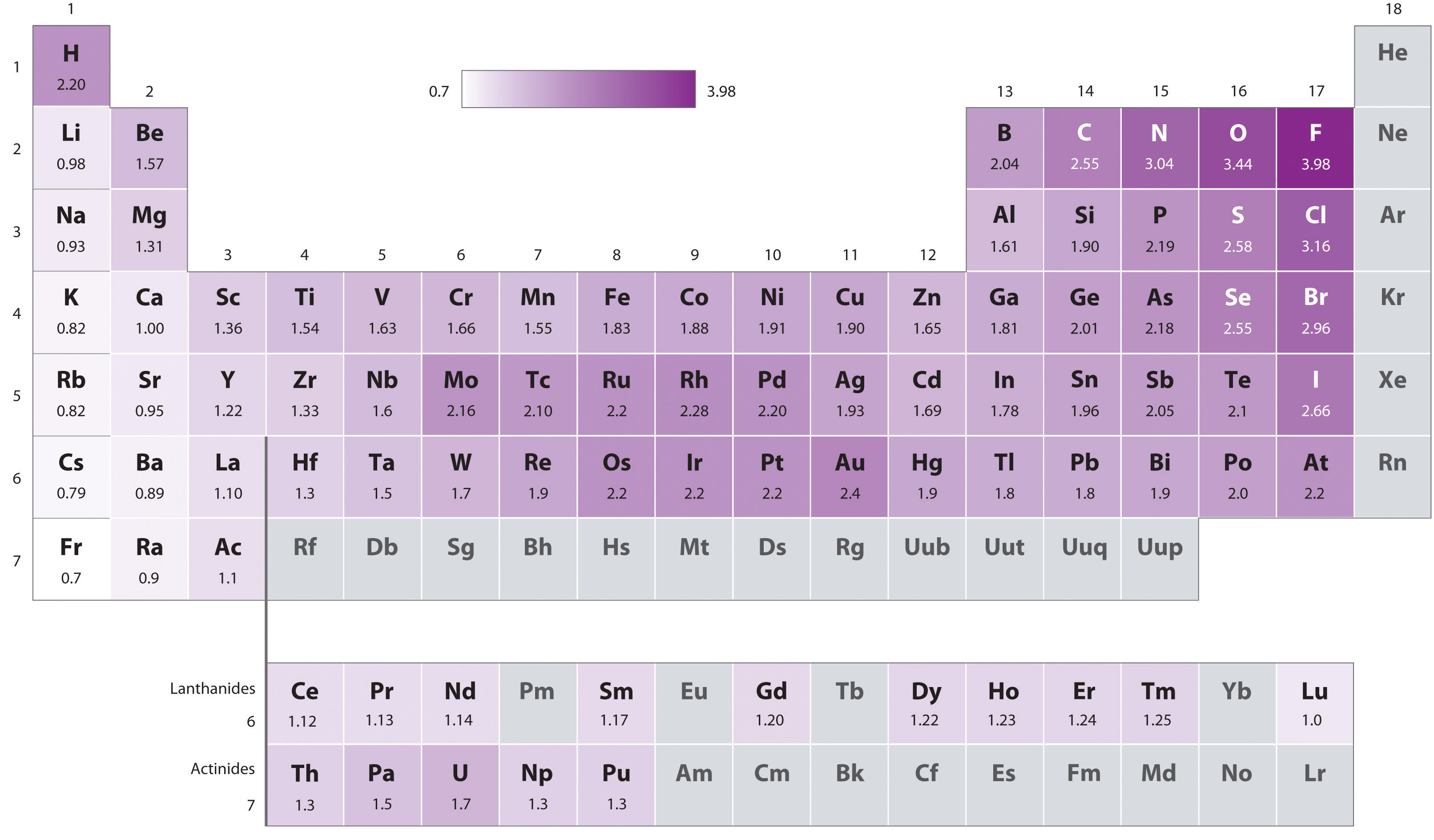
WCAG 2.0 contains 12 guidelines organized under 4 principles: Perceivable, Operable, Understandable, and Robust (POUR for short). Arrange these elements according to electronegativity: Rb N O K Ga Arrange these elements in order of decreasing electronegativity: Rank from largest to smallest. Arrange these elements according to electronegativity. This certifies it as a stable and referenceable technical standard. WCAG 2.0 is also an international standard, ISO 40500. The elements are placed in table cells, in reading order of ascending atomic number.The table columns are called groups, the rows are called periods.The breaks at the end of each period occur according to a repetition (or periodicity) of physical and chemical properties of the elements. This website adopts the Web Content Accessibility Guidelines (WCAG 2.0) as the accessibility standard for all its related web development and services. The periodic table is a 2-dimensional structured table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
